What Is Electrical Conductivity In Metals: Exploring Electrical Flow
Which Metals Conduct Electricity The Best? | Metal Supermarkets
Keywords searched by users: What is electrical conductivity metals Electrical conductivity of metals, electrical conductivity of metals pdf, What is electrical conductivity, electrical conductivity of metals formula, electrical conductivity of metals with temperature, electrical conductivity of metals chart, electrical conductivity of metals in increasing order, Best conductive metal
What Is Electrical Conduction In Metals?
Electric conduction in metals is a phenomenon where electric current flows through a metal material due to the movement of free electrons between the metal atoms. Unlike electrons in covalent bonds, these free electrons are not bound to any specific atom. This fundamental characteristic of metals, which enables them to conduct electricity effectively, is a result of the unique arrangement of electrons in the metal’s atomic structure. This property plays a crucial role in various applications, from wiring and electrical circuits to industrial processes and electronic devices. It’s important to note that metals are not the only materials capable of conducting electricity, but their exceptional conductivity is a key feature that distinguishes them in the realm of electrical conduction.
What Is Meant By Electrical Conductivity?
Understanding Electrical Conductivity
Electrical conductivity refers to the intrinsic property of a material that determines its ability to facilitate the flow of electric current. In essence, it quantifies how easily a material permits the passage of electricity. This property varies from one substance to another, as it hinges on their unique capacity to allow the unhindered movement of electric charges. Consequently, materials can be categorized based on their electrical conductivity, from excellent conductors like metals to insulators, which impede the flow of electric current. In summary, electrical conductivity serves as a crucial parameter for characterizing and distinguishing different materials based on their electrical behavior.
Update 44 What is electrical conductivity metals

:max_bytes(150000):strip_icc()/the-most-conductive-element-606683_FINAL-cb8d31a0404241e2a3187e67c7b57e8c.gif)

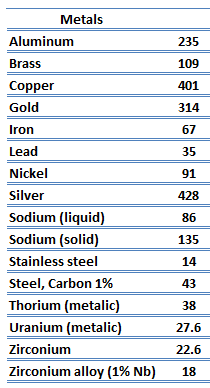

Categories: Details 13 What Is Electrical Conductivity Metals
See more here: g3magazine.com

The property by which metals facilitate the flow of electric current through them is called electrical conductivity. 2. All metals are good conductors of electricity because they contain free or mobile electrons. These free electrons conduct electric current.Metals conduct electricity by allowing free electrons to move between the atoms. These electrons are not associated with a single atom or covalent bond.What is Electrical Conductivity? Electrical conductivity is nothing but the measure of the capability of the material to pass the flow of electric current. Electrical conductivity differs from one material to another depending on the ability to let the electricity flow through them.
Learn more about the topic What is electrical conductivity metals.
- Explain electrical conductivity of metals. [3 MARKS] – BYJU’S
- What Is Conductivity in Metals? – MetalTek International
- Conductivity of Water and Saltwater, Specific Conductance, FAQs – BYJU’S
- Electrical Conductivity | Definition, Formula & Examples
- Why are metals good conductors of electricity? – Toppr
- Electrical and Thermal Conductivity – TECHNICALTIDBITS